Comparing electrochemical perormance of transition metal silicate cathodes and chevrel phase Mo6S8 in the analogous rechargeable Mg-ion battery system
Journal article, Peer reviewed
Accepted version
Permanent lenke
http://hdl.handle.net/11250/2472667Utgivelsesdato
2016Metadata
Vis full innførselSamlinger
- Institutt for materialteknologi [2549]
- Publikasjoner fra CRIStin - NTNU [38127]
Sammendrag
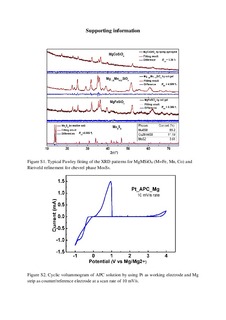
Polyanion based silicate materials, MgMSiO4 (M = Fe, Mn, Co), previously reported to be promising cathode materials for Mg-ion batteries, have been re-examined. Both the sol-gel and molten salt methods are employed to synthesize MgMSiO4 composites. Mo6S8 is synthesized by a molten salt method combined with Cu leaching and investigated in the equivalent electrochemical system as a bench mark. Electrochemical measurements for Mo6S8 performed using the 2nd generation electrolyte show similar results to those reported in literature. Electrochemical performance of the silicate materials on the other hand, do not show the promising results previously reported. A thorough study of these published results are presented here, and compared to the current experimental data on the same material system. It appears that there are certain inconsistencies in the published results which cannot be explained. To further corroborate the present experimental results, atomic-scale calculations from first principles are performed, demonstrating that diffusion barriers are very high for Mg diffusion in MgMSiO4. In conclusion, MgMSiO4 (M = Fe, Mn, Co) olivine materials do not seem to be such good candidates for cathode materials in Mg-ion batteries as previously reported.